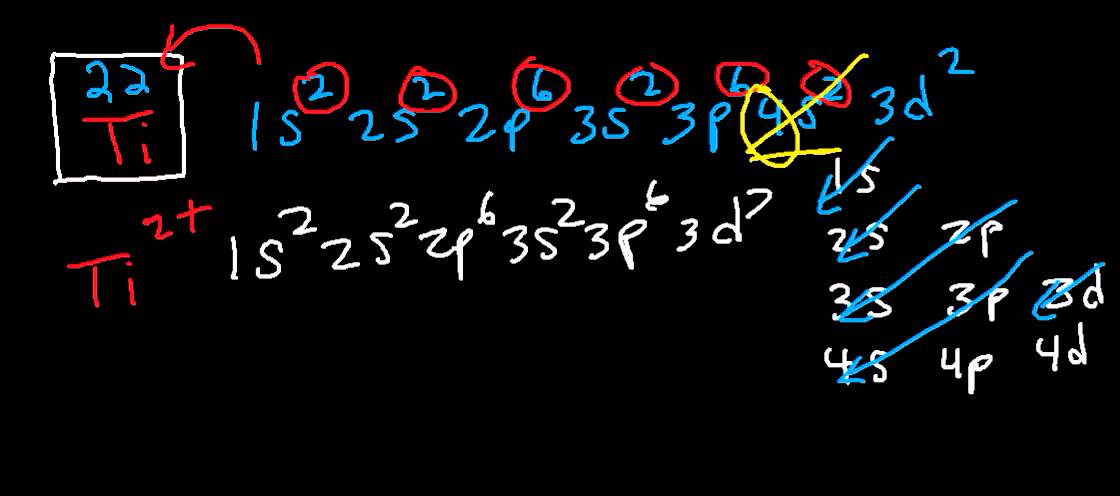Electronic states of neutral and cationic bis(benzene) titanium and vanadium sandwich complexes studied by pulsed field ionization electron spectroscopy: The Journal of Chemical Physics: Vol 127, No 11

SOLVED: Which of the following ions have noble gas electron configurations? (Select all that apply: (a) Y3+, Mn4+, Fe 3+ V2+ Y3+ Mn4+ Fe3+ vz+ (b) T1+ , Ti3+, 02- TI+ TI3+

Notes 4 ions - CHEM 1a Energy Levels in Many-Electron Atoms The following figure shows the electron - Studocu

Electron Configuration for Ti , Ti3+, and Ti4+ (Titanium and Titanium I... | Electron configuration, Electrons, Educational videos
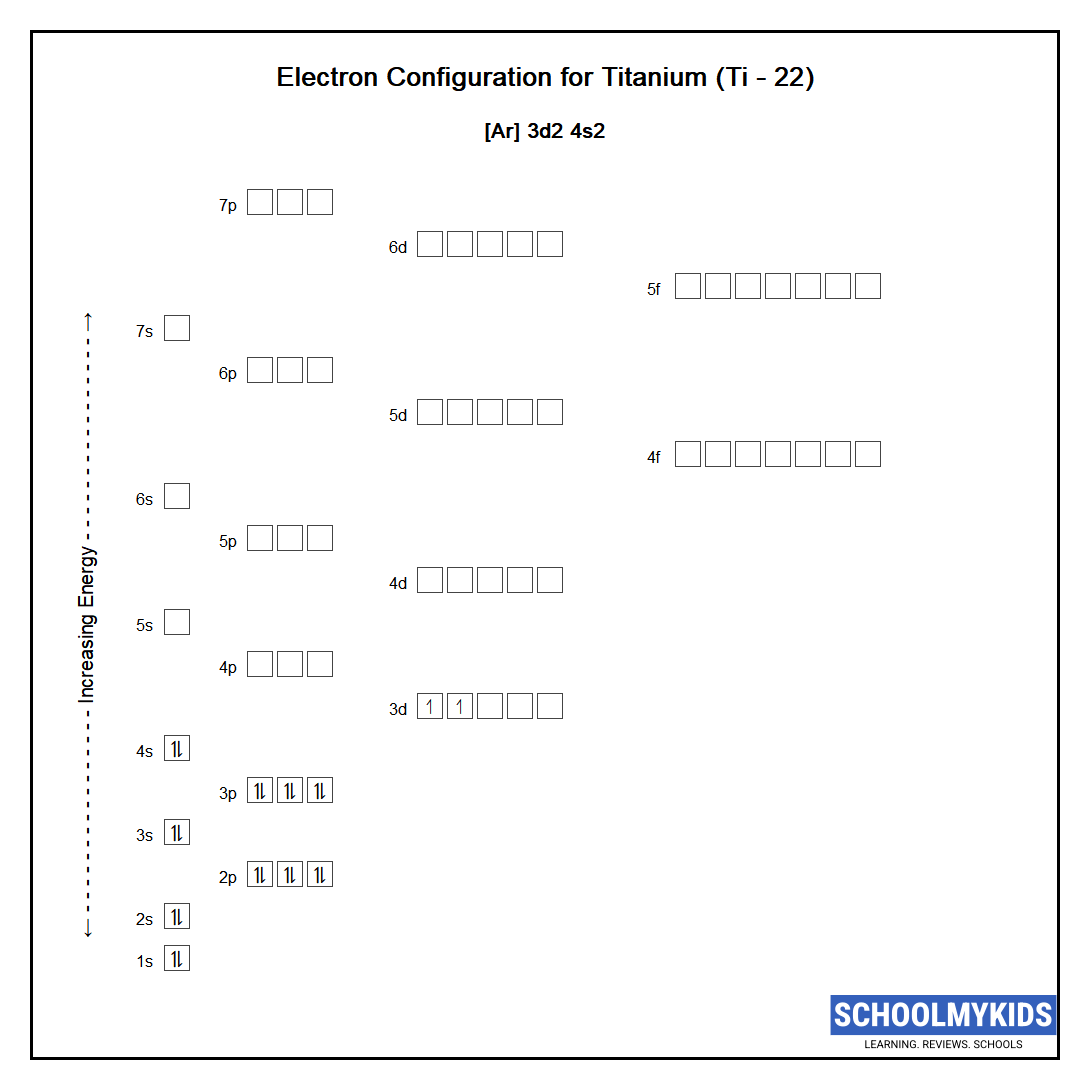
Ti Titanium Element Information: Facts, Properties, Trends, Uses and comparison - Periodic Table of the Elements | SchoolMyKids
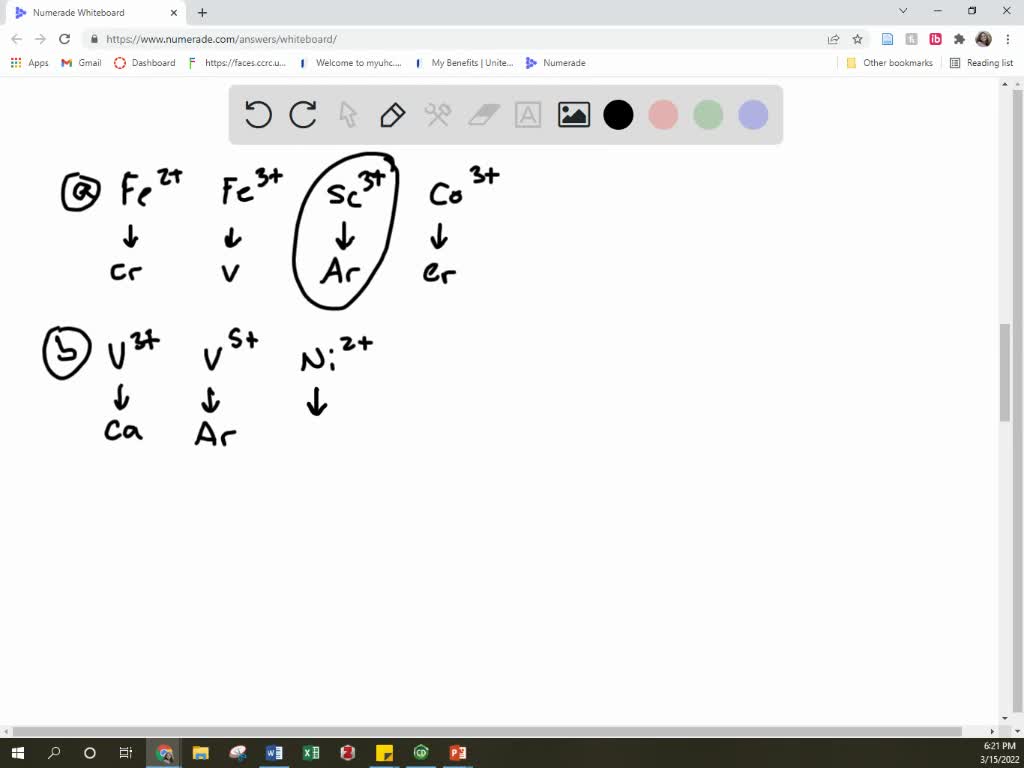
SOLVED: Which of the following ions have noble gas electron configurations? (a) Fe2+ Fe3+ Sc3+, Co3+ chemPad Help XoXi Cleel (b) v3+ , VS+ , Ni2+ chcmPac Help Xeki celt (c) Pu4+

Reactions of Th+ + H2, D2, and HD Studied by Guided Ion Beam Tandem Mass Spectrometry and Quantum Chemical Calculations | The Journal of Physical Chemistry B