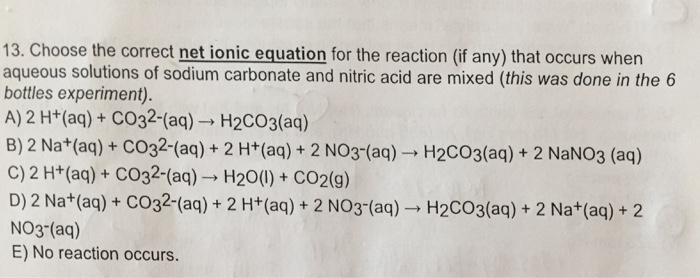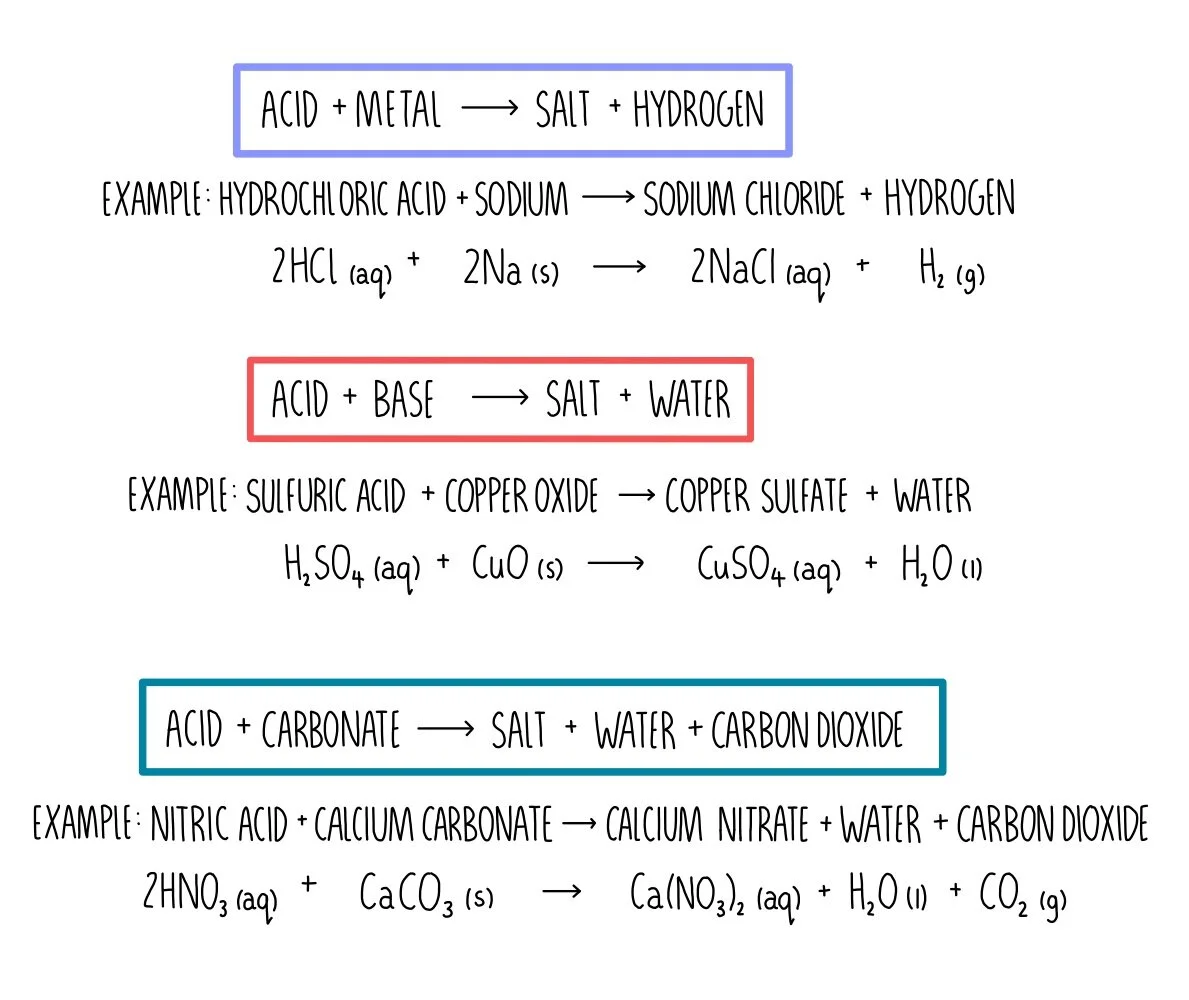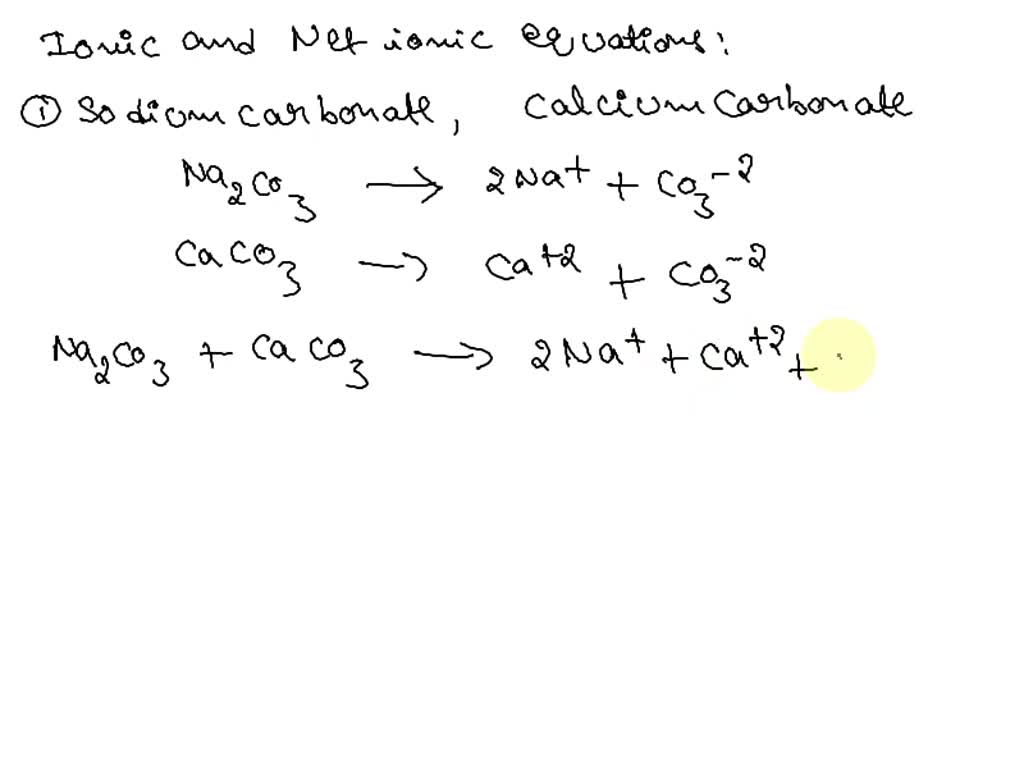
SOLVED: write the molecular equation, total ionic equation, net ionic equation for the following1. lithium carbonate + nitric acid2. calcium nitrate + potassium carbonate 3. phosphoric acid + calcium hydroxide

SOLVED: Question 7 (1 point) Sodium carbonate reacts with nitric acid to form sodium nitrate; carbon dioxide, and water according to the balanced reaction below How many grams of sodium carbonate are
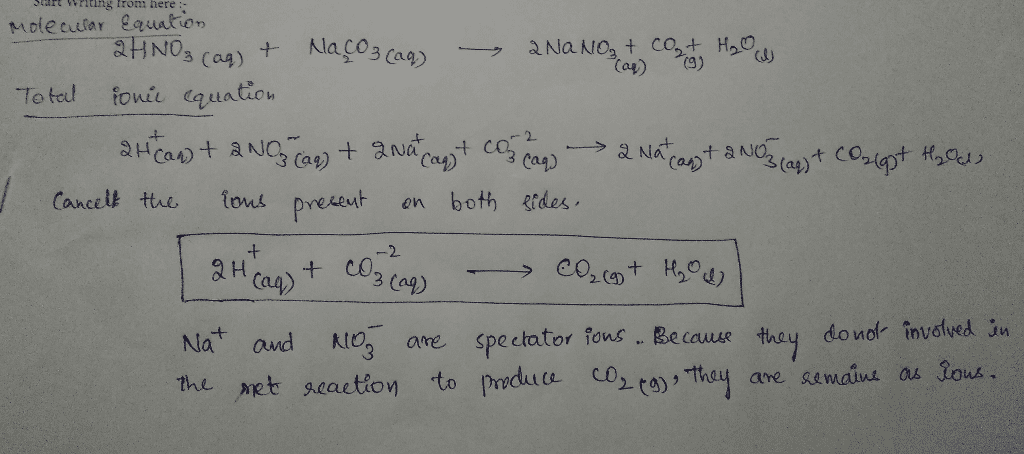
OneClass: Write a net ionic equation for the reaction that occurs when excess nitric acid (aq) and so...

H 2 SO 4 + Zn 1) Sulphuric acid + zinc 3) Nitric acid + sodium thiosulphate 2) Hydrochloric acid + magnesium 4) Hydrogen peroxide with catalyst Popular. - ppt download
Write balanced chemical equation for the following : 1. Reaction of nitric acid with sodium bicarbonate. - Sarthaks eConnect | Largest Online Education Community


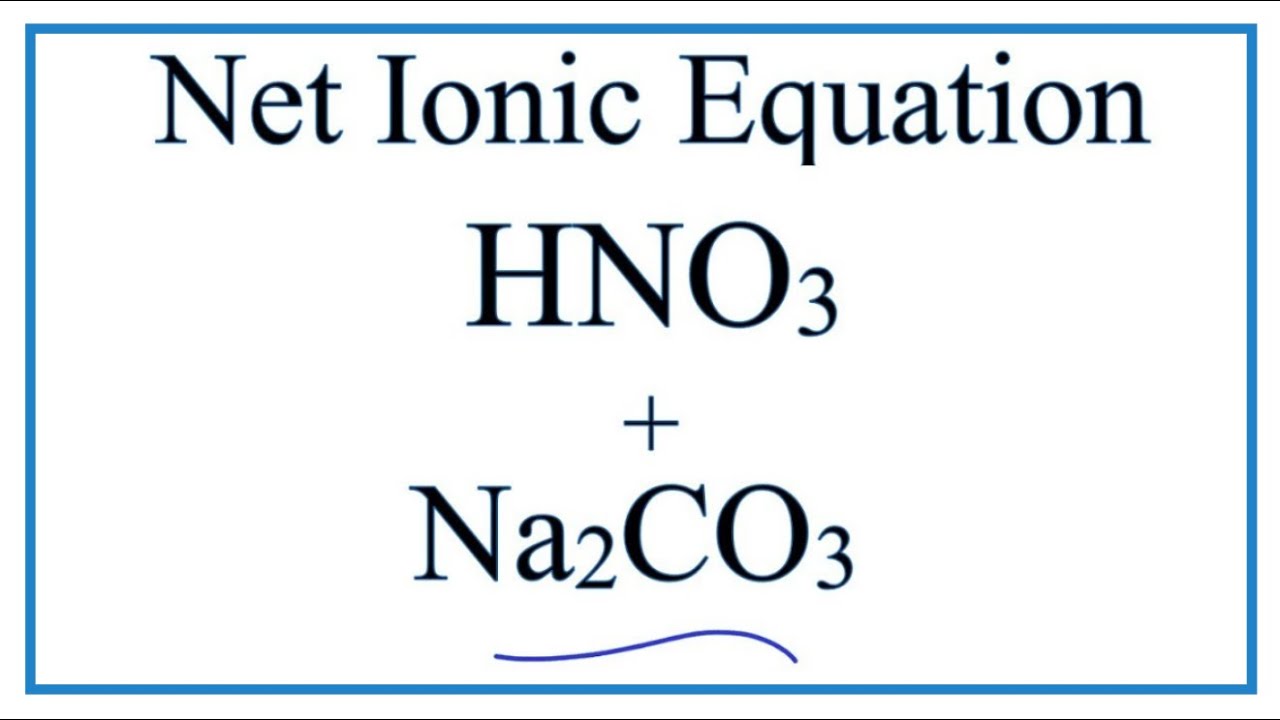
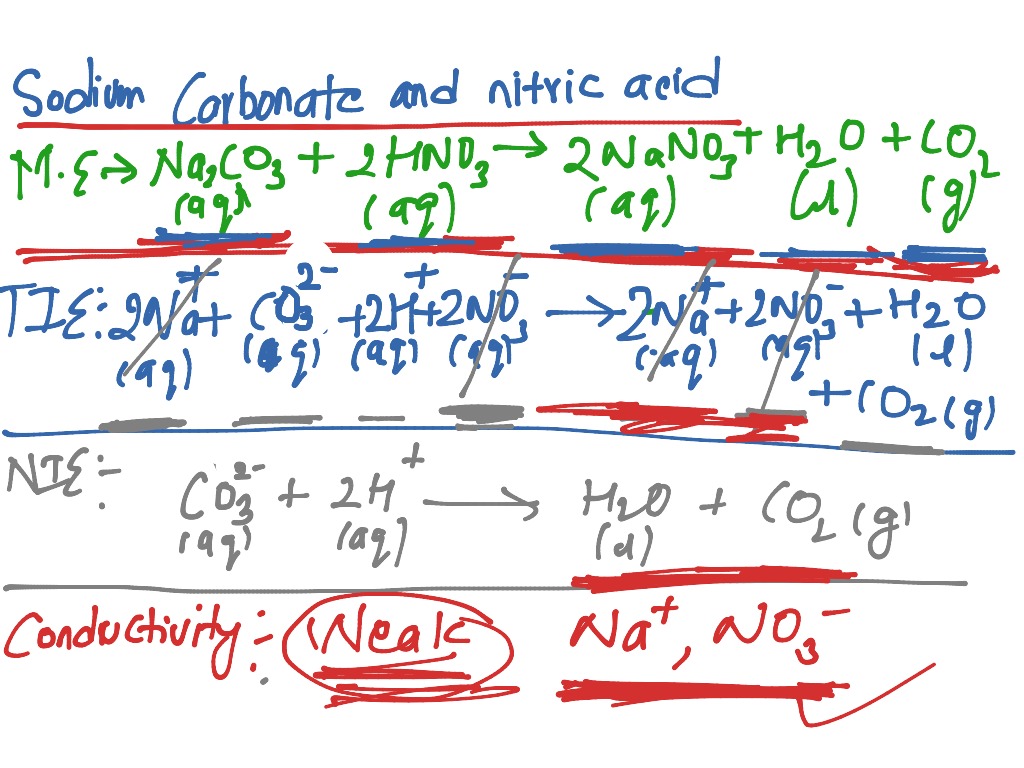





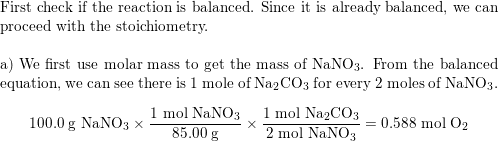
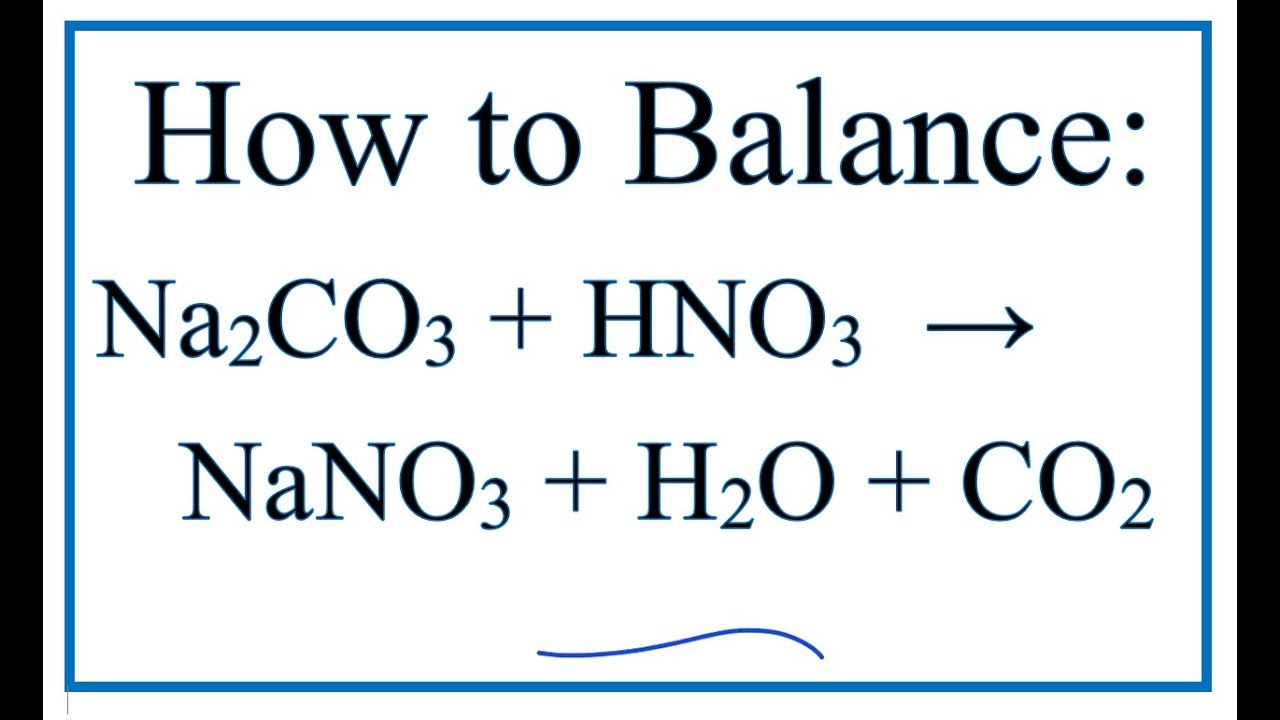
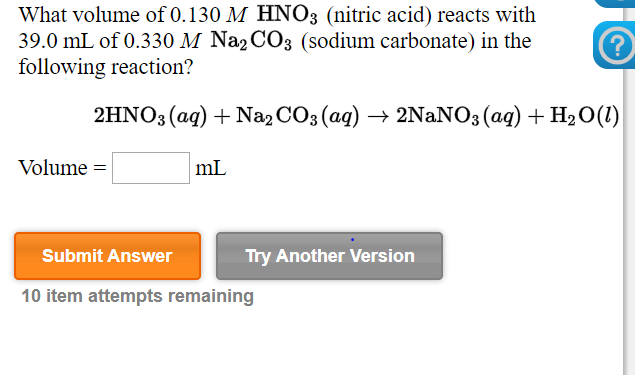



![ANSWERED] When nitric acid (HNO3) reacts with sodium... - Organic Chemistry ANSWERED] When nitric acid (HNO3) reacts with sodium... - Organic Chemistry](https://media.kunduz.com/media/sug-question/raw/59811332-1659634030.9246697.jpeg)