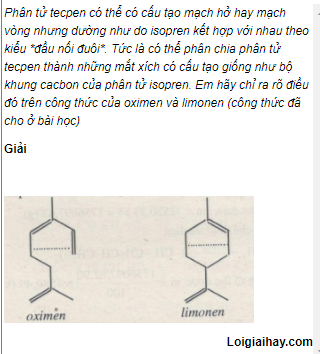
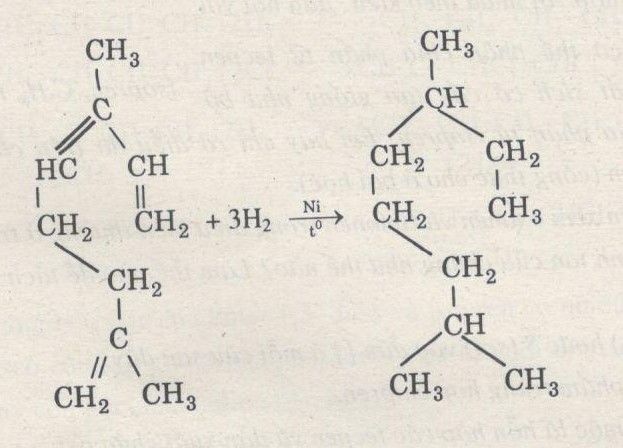
Beckmannsche Umlagerung bei o‐ und p‐Chinon‐oximen - Beckmann - 1923 - Berichte der deutschen chemischen Gesellschaft (A and B Series) - Wiley Online Library
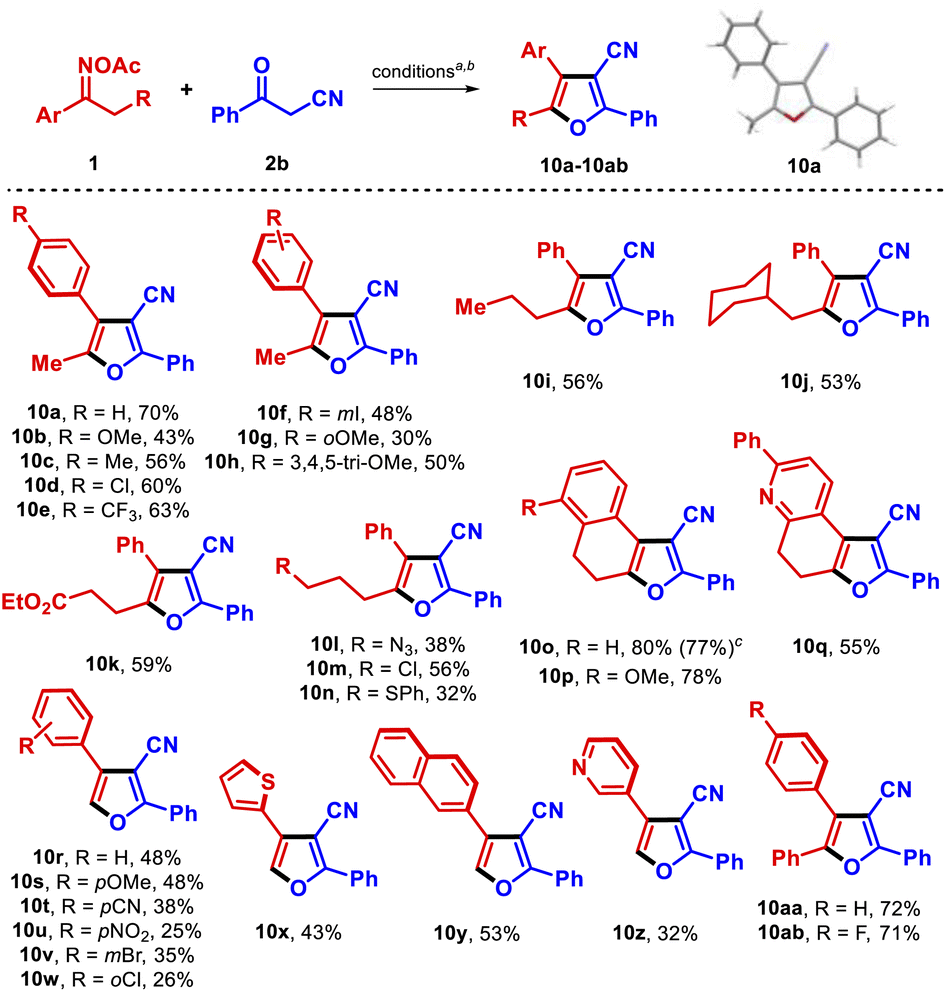
Divergent copper-catalyzed syntheses of 3-carboxylpyrroles and 3-cyanofurans from O -acetyl oximes and β-ketoesters/nitriles - RSC Advances (RSC Publishing) DOI:10.1039/D2RA04938D

Palladium‐Catalyzed Dearomative syn‐1,4‐Oxyamination - Tang - 2019 - Angewandte Chemie - Wiley Online Library

Unusual Domino Michael/Aldol Condensation Reactions Employing Oximes as N‐Selective Nucleophiles: Synthesis of N‐Hydroxypyrroles - Tan - 2009 - Angewandte Chemie - Wiley Online Library

Intermolecular Cope‐Type Hydroamination of Alkenes and Alkynes - Beauchemin - 2008 - Angewandte Chemie - Wiley Online Library

Über die Absorptionsspektren von Oximen - Meisenheimer - 1933 - Justus Liebigs Annalen der Chemie - Wiley Online Library

![PDF] Freisetzung von Stickstoffmonoxid aus Oximen und Dephostatin-Derivaten | Semantic Scholar PDF] Freisetzung von Stickstoffmonoxid aus Oximen und Dephostatin-Derivaten | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ddbea379c3e8ace1552a8047ab54389379553ba8/165-Figure56-1.png)
![PDF] Freisetzung von Stickstoffmonoxid aus Oximen und Dephostatin-Derivaten | Semantic Scholar PDF] Freisetzung von Stickstoffmonoxid aus Oximen und Dephostatin-Derivaten | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ddbea379c3e8ace1552a8047ab54389379553ba8/167-Figure57-1.png)