Formation of copper oxide nanoparticles over silica by means of the... | Download Scientific Diagram

Stability of Residual Oxides in Oxide‐Derived Copper Catalysts for Electrochemical CO2 Reduction Investigated with 18O Labeling - Lum - 2018 - Angewandte Chemie International Edition - Wiley Online Library
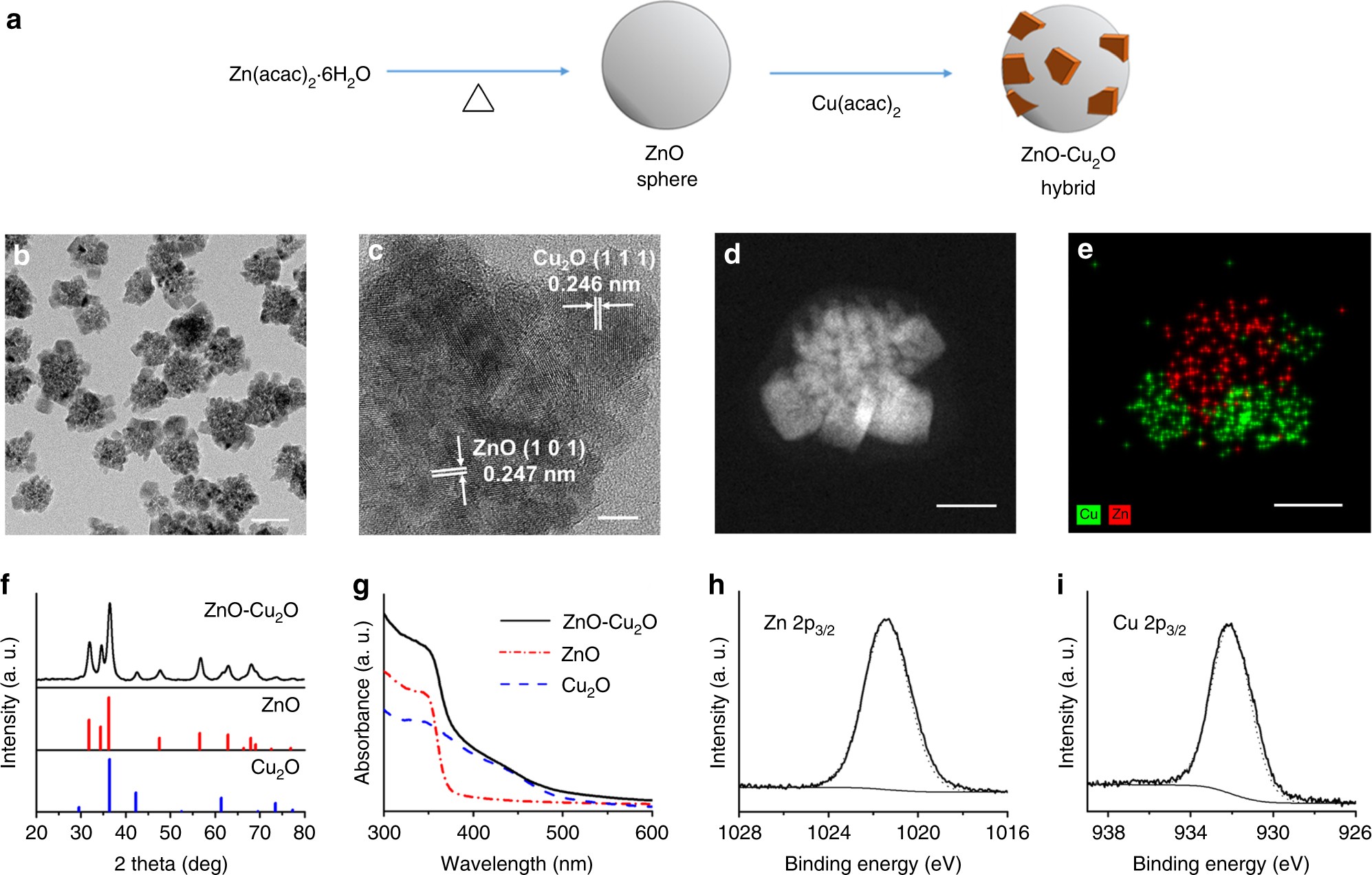
Colloidal zinc oxide-copper(I) oxide nanocatalysts for selective aqueous photocatalytic carbon dioxide conversion into methane | Nature Communications

Practical methods of organic chemistry . l at its upper end. Intothe tube lying in a horizontal positionthe copper oxide spiral is pushed sofar that its loop is 5 cm. from