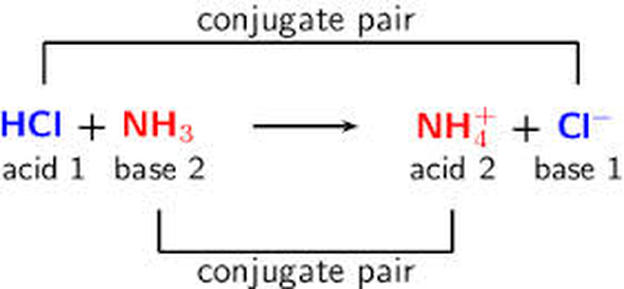
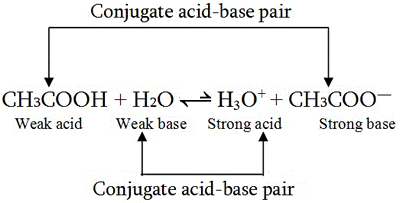
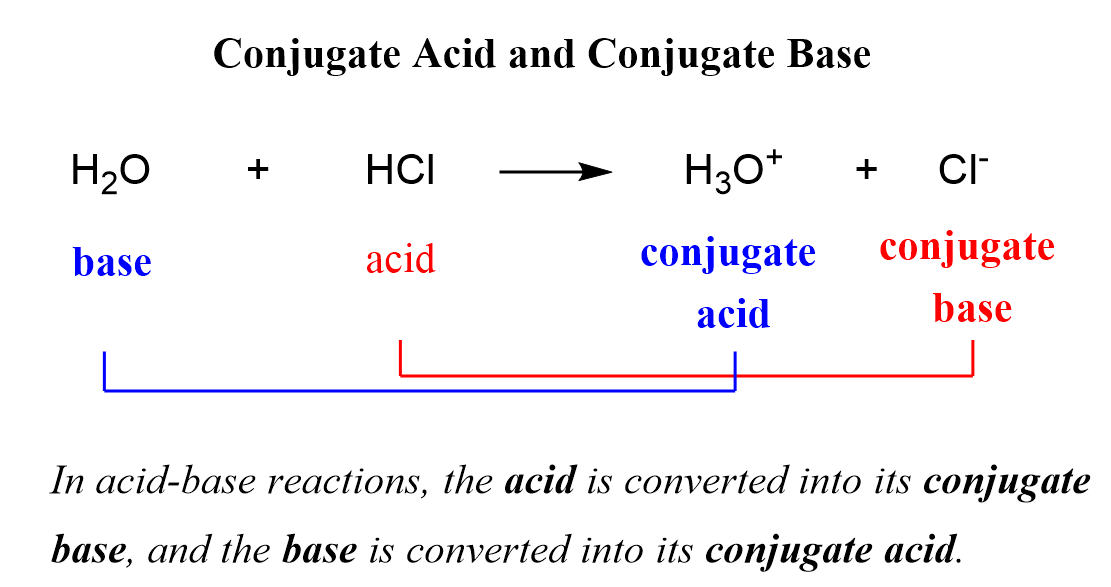
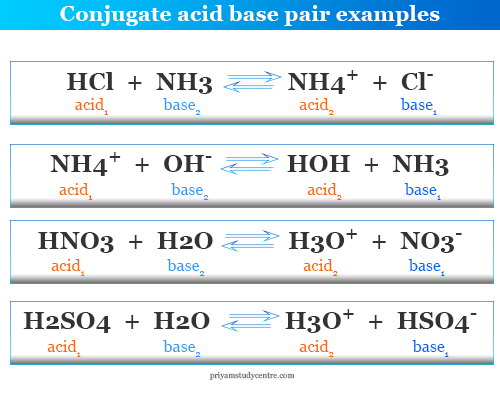
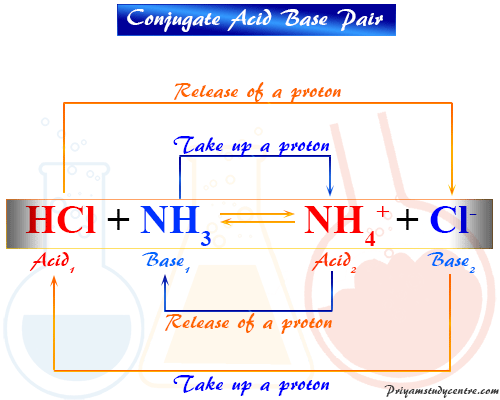
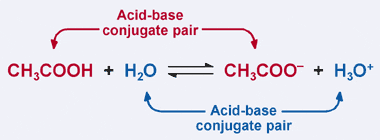
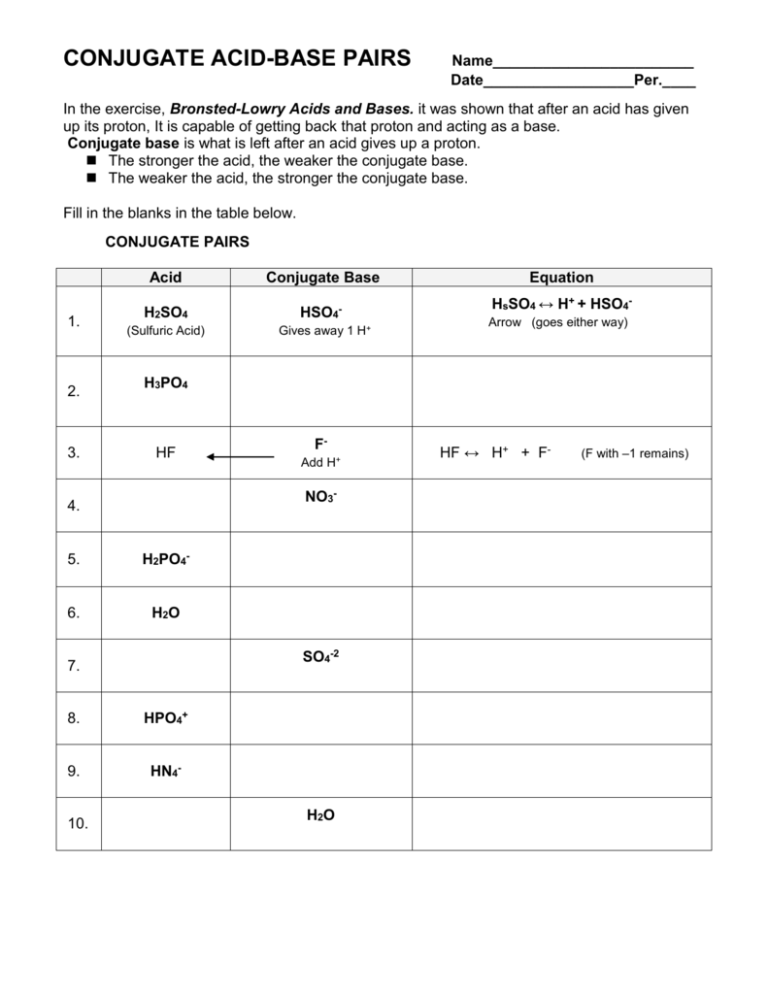
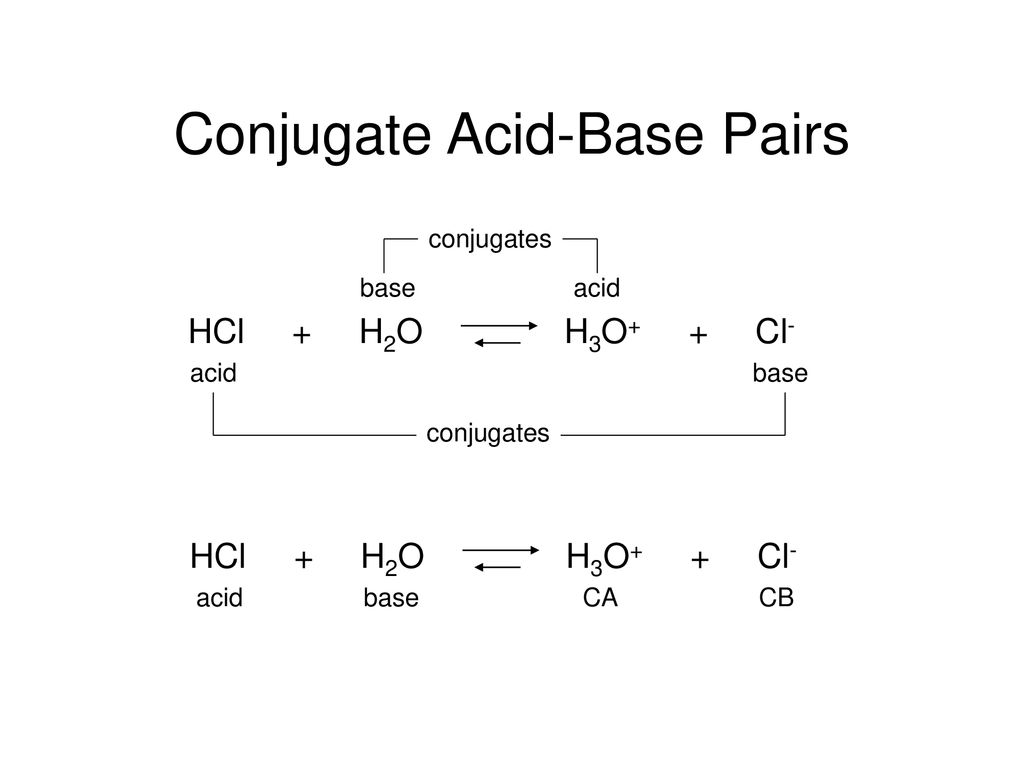
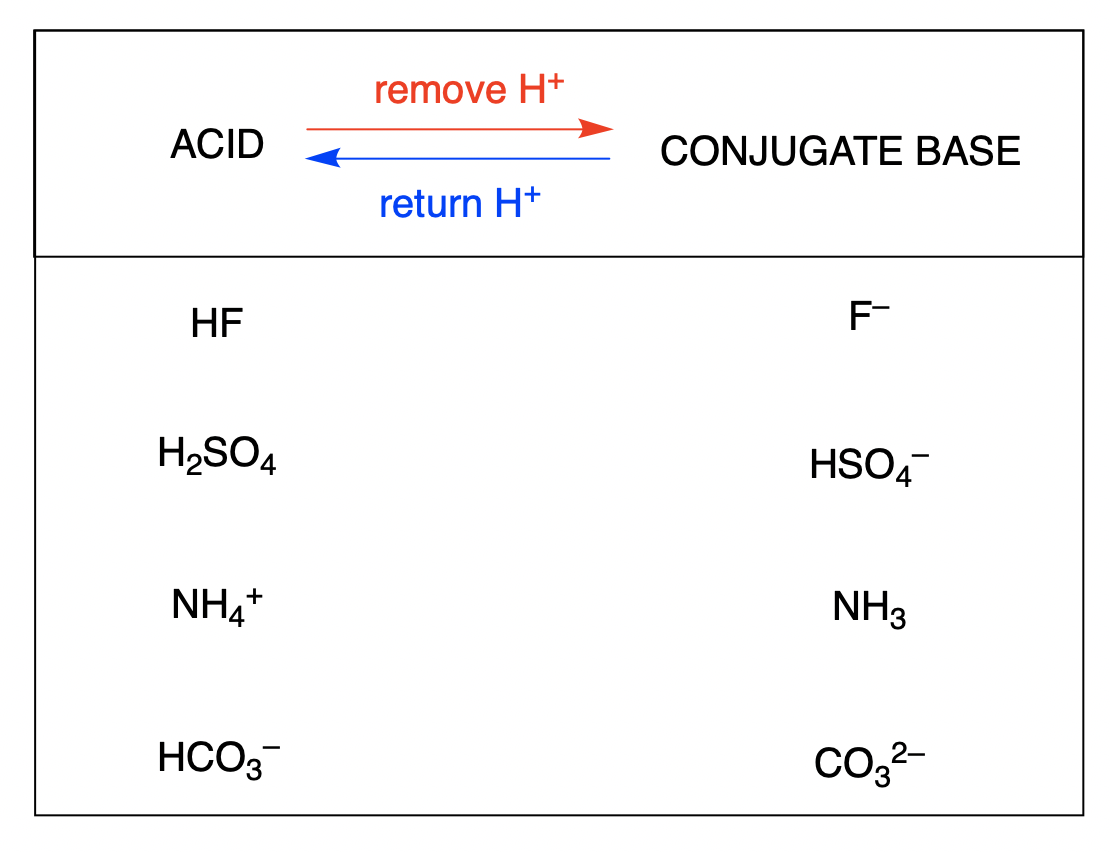
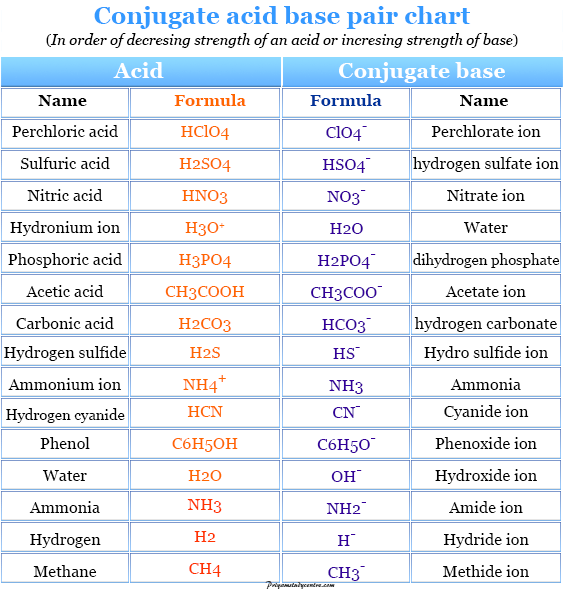
What is meant by the conjugate acid - base pair? Find the conjugate acid/ base for the following species. HNO2,CN^-,HClO4F^-,OH^-,CO3^2 - and S^2 -

What do you understand by conjugate acid base pairs? Give examples. from Chemistry Equilibrium Class 11 Karnataka Board

SOLVED: 4. Which of the following is a conjugate acid/base pair? (3 Points) H3O+ /OH HCl/OCL - HzSO4/S04 NHt /NH3 noneofthese