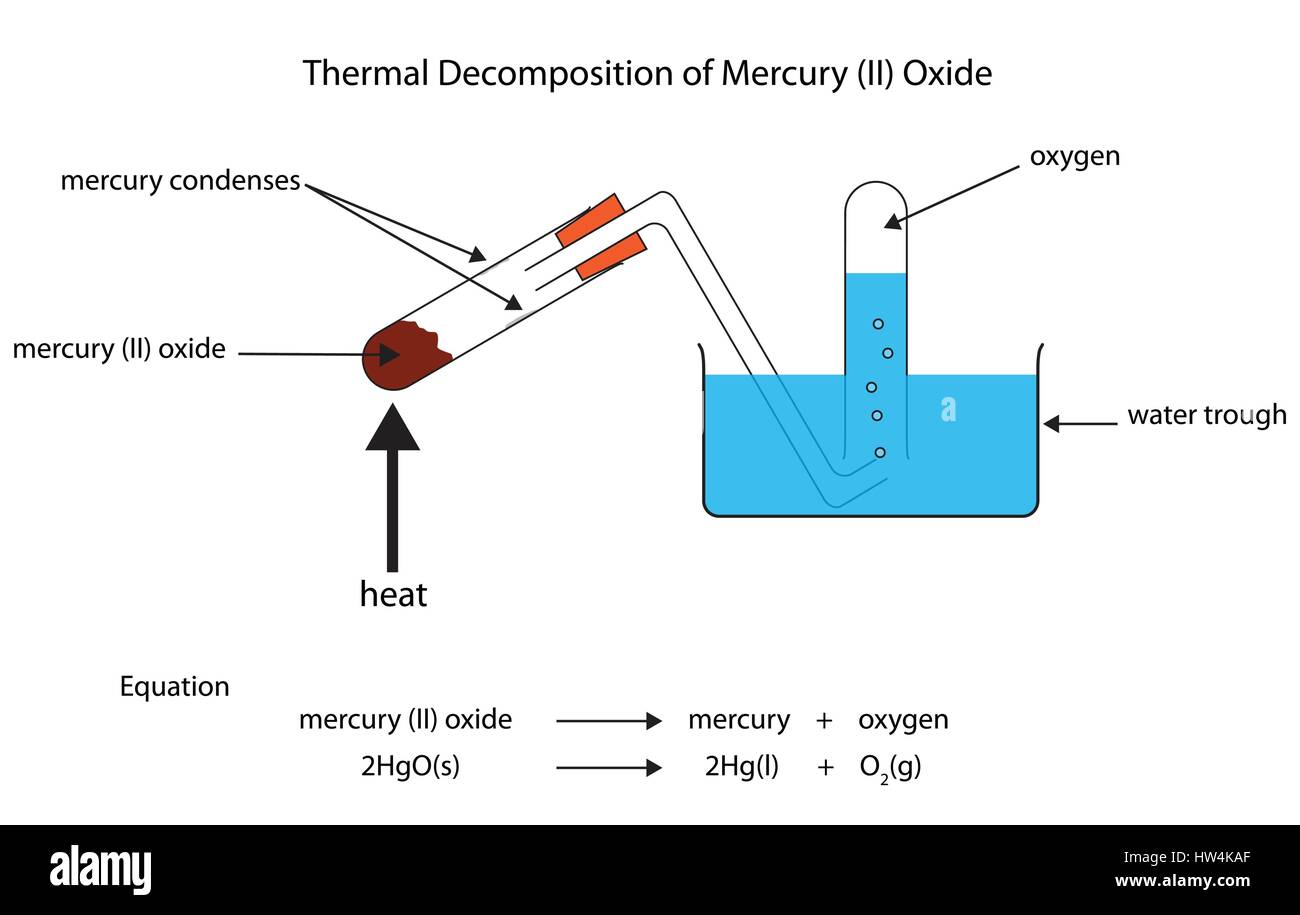
Which gas is produced when mercury oxide is heated in a hard glass tube? How do you test the presence of this gas? - Quora

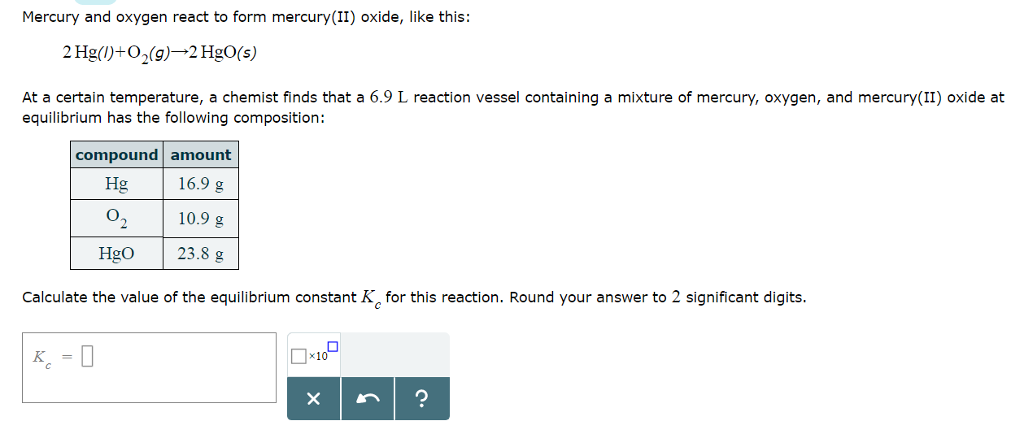
According to the law of conservation of mass, how much mercury was present in the mercury (II) oxide? - Brainly.com

SOLVED: unds con't Mercury (II) oxide Heat Mercury and oxygen Decomposition reactions: 2 HgO + Heat 2Hg 02 Sodium Bicarbonate + Heat sodium carbonate + water + carbon dioxide 2NaHCO3 Heat Naz